INTRODUCTION
Cauda equina syndrome (CES), which is mainly caused by severe compression of the nerve roots below the conus medullaris, is one of the most serious and complicated spinal pathologies. It is a relatively rare condition most commonly caused by extreme lumbar disc herniation (LDH) and accounts for about 1% to 3% of LDH patients [1,2]. This syndrome causes characteristic symptoms including saddle anesthesia, bowel or bladder dysfunction, sexual dysfunction, severe lower back pain, unilateral or bilateral sciatica, and motor weakness [1,3]. It is regarded as an emergent condition and it is widely acknowledged that a prompt diagnosis and urgent decompression are critical for better neurological outcomes [4,5].
Traditionally, open lumbar microdiscectomy (OLM) has been widely considered as a standard procedure for CES [6-9]. Surgical techniques of OLM are continually evolving, with a trend toward less aggressive, less invasive procedures to reduce surgical stress on the patient [10-14]. OLM provides additional decompression of nerve roots via laminectomy; however, OLM results in muscle damage, clinically symptomatic scarring of the epidural space, increased risk of postoperative spinal instability, and chronic back pain [15-17]. Moreover, due to the innate nature of the posterior approach, it requires nerve root retraction during discectomy, which increases the risk of neural damage, especially in the case of CES with a huge central disc rupture [18-20].
With the instrumental development of endoscopes and in an attempt to reduce the complication rate, percutaneous endoscopic lumbar discectomy (PELD) has become increasingly popular over the past years. Many studies have shown comparable clinical outcomes of PELD compared with OLM [21-24]. PELD also has several advantages over OLM, including less soft tissue trauma, better bony preservation and rapid recovery [25]. In particular, PELD has the advantage of avoiding root retraction which is evitable in OLM.
On the basis of these advantages, several studies have reported favorable outcomes of CES treated by PELD [26-28]. However, to the best of our knowledge, no study has compared the clinical outcome of CES using PELD and OLM. In this retrospective cohort study, we compared the clinical outcomes when using PELD and OLM to treat CES.
MATERIALS AND METHODS
1. Patient Population
We performed a retrospective cohort study approved by the Institutional Review Board of Daegu Wooridul Spine Hospital (IRB No. 2022-01-WSH-001), and all participants gave informed consent before enrollment. Between January 2017 and Fifteen patients with CES underwent PELD or OLM.December 2019, a total of 15 consecutive patients with CES caused by lumbar central huge disc rupture were treated in Daegu Wooridul Spine Hospital by 4 neurosurgeons. Each surgeon had performed over 300 cases of PELD and over 1,000 cases of OLM throughout their career.
Inclusion criteria were a single level central huge LDH with the following so-called “red flag” symptoms: (1) unilateral or bilateral motor weakness, (2) absent or decreased perineal sensation, (3) absent or decreased or anal sphincter tone, or (4) bladder dysfunction. Exclusion criteria were as follows: (1) LDH concomitant with spinal stenosis, (2) disc herniation with calcified disc, (3) instability, (4) epidural abscess, or (5) neoplasms.
All cases were treated by PELD (with 7 patients, 5 males and 2 females) or OLM (with 8 patients, 6 males and 2 females) within a day of diagnosis of CES and postoperative magnetic resonance imaging was obtained right after the patients were allowed to stand and walk independently.
Medical chart and image databases were analyzed. Patient demographics including age, sex, affected level, body mass index (kg/m2), duration of symptoms before treatment, and neurological symptoms of CES were reviewed.
2. Surgical Techniques
1) Percutaneous Endoscopic Lumbar Discectomy
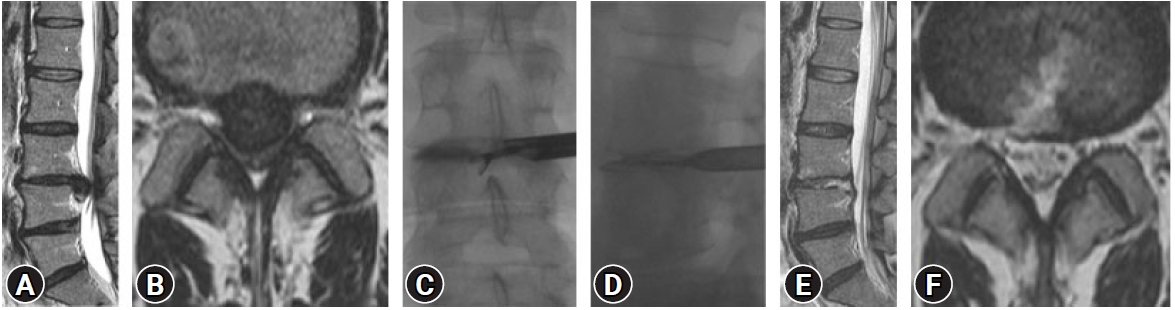
A standard transforaminal PELD procedure was performed using the inside-out technique. The procedure was performed under local anesthesia with the patient in the prone position on a radiolucent table and receiving supplemental nasal oxygen. An imaginary line was drawn to the annular puncture site and the skin entry site was marked for the planned surgical trajectory. After infiltration of the entry point (10–12 cm from the midline) with local anesthetics, an 18-gauge spinal needle was introduced into Kambin’s triangle under the fluoroscopic guidance with continuous patient feedback. The final target point of the spinal needle was the medial pedicular line on the anteroposterior view and posterior vertebral line on the lateral view. After inserting the needle in to the disc, discography using indigo carmine was performed to distinguish the pathological fragment clearly during the procedure. After insertion of a guide wire through the spinal needle, the spinal needle was removed and a small skin incision at the entry point was made. A tapered cannulated obturator was inserted along the guide wire and after contacting the annulus, the obturator was inserted into the disc space with hammering until its tip reached the midline on the anteroposterior view. A beveled working cannula was inserted into the disc space along the obturator under fluoroscopic guidance. After removing the obturator, an endoscope (TESSYS System; Joimax, Karlsruhe, Germany) was inserted through the working cannula and positioned at the annular defect site. This was confirmed using axial magnetic resonance imaging preoperatively. A targeted fragmentectomy was performed and constant saline irrigation was administered throughout the whole procedure. To remove the trapped disc fragment, the annular defect site was widened using a side-firing holmium: YAG laser (Lumenis Inc., Yokneam, Israel). In cases where the disc fragment was too large to pass through the cannula, it was vaporized by a laser or bipolar radiofrequency coagulator (Ellman International, Hicksville, NY, USA) to reduce the volume and removed with forceps. After removal of the disc fragment and decompression, the beating of the traversing nerve root and dural sac with the pulse of the artery was confirmed. The endpoints of neural decompression were complete visualization of the dural sac and traversing root, dural pulsation, irrigation flutter, and cough impulse. After confirming the relief of preoperative symptoms by asking the patients, the endoscope was withdrawn, and a sterile dressing was applied with a one-point suture (Figure 1).
2) Open Lumbar Microdiscectomy
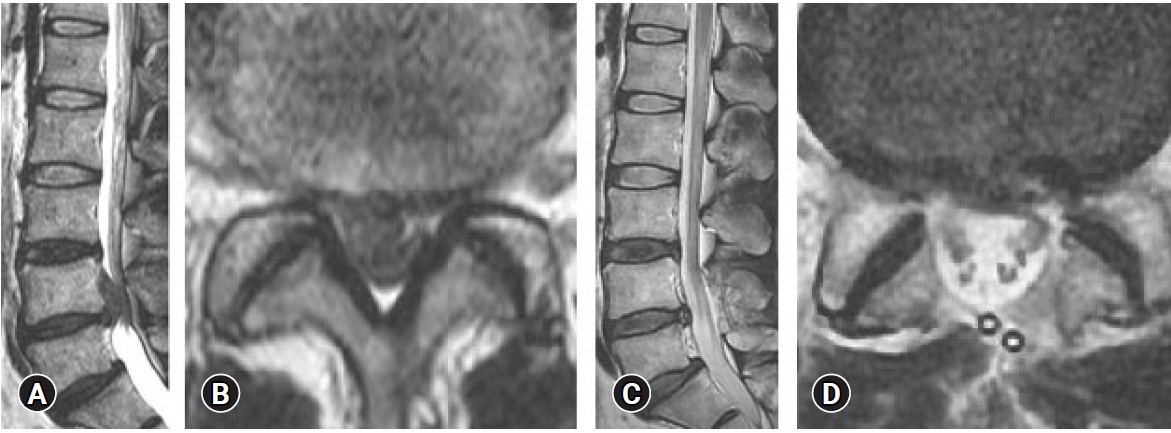
The patient was placed in a kneeling prone position under general anesthesia. After confirming the target level using fluoroscopy, a 3 cm midline longitudinal skin incision was made, and the paravertebral muscles were dissected and retracted laterally. A Caspar lumbar retractor was applied to obtain a direct view of the operating field and the operative level was confirmed by fluoroscopy. OLM was performed following a bilateral approach. Under microscopic visualization, partial hemilaminectomy, medial facetectomy, and foraminotomy using a high-speed drill were performed. After removal of the ligamentum flavum, the same procedure was performed on the opposite side. The nerve root and thecal sac were retracted gently and the herniated disc fragment was removed with pituitary forceps. Following discectomy, the thecal sac and root were pulsated and retracted without resistance, confirming adequate neural decompression. After meticulous bleeding control, the muscle, subcutaneous tissue and skin were sutured in layer (Figure 2)
3. Statistical Analysis
All statistical analyses were performed using SPSS Version 25 (IBM Corporation, Armonk, NY, USA). Quantitative data were expressed as the mean±SD (standard deviation) or frequency. Each category and difference between two groups were compared using appropriate statistical tools such as the Pearson correlation, Fisher’s exact test, the chi-square test, or the Mann-Whitney U-test. A p-value of <0.05 was considered statistically significant.
RESULTS
A total of 15 consecutive hospitalized patients with CES caused by lumbar central huge disc rupture were treated Daegu Wooridul Spine Hospital. Of the 15 patients, 7 underwent PELD and the remaining 8 underwent OLM. On postoperative magnetic resonance imaging, the disc fragment was removed completely in all cases. All patients were followed-up over a year after the procedure in an outpatient clinic and their clinical outcomes were recorded in detail on a medical chart. One patient in each group had a telephone survey because they refused to visit the hospital.
The patients’ demographic and clinical characteristics are summarized in Table 1. There were no significant differences in preoperative demographic characteristics between the PELD and OLM groups (p>0.05).
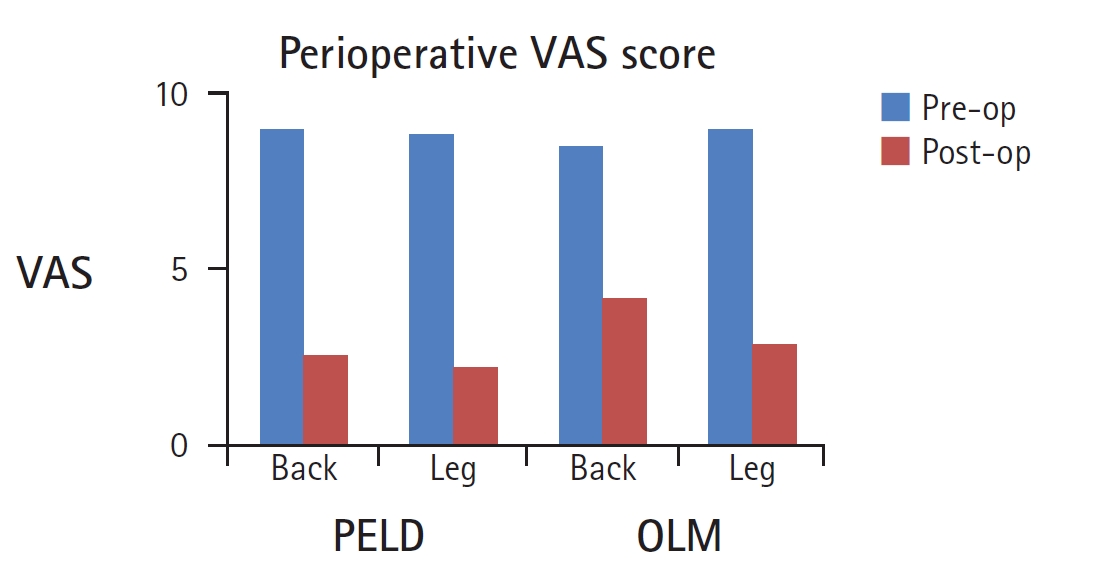
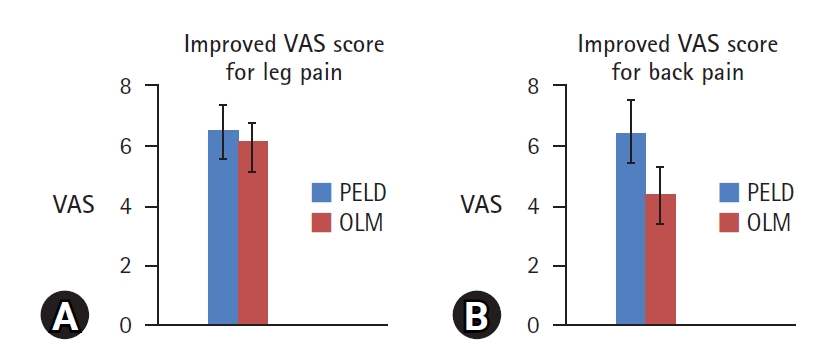
The perioperative outcomes of PELD and OLM for CES are summarized in Table 2. Between the two groups, there were no significant differences in preoperative VAS score divided into back and radiating leg pain (p>0.05). Both groups reported a significant postoperative reduction of VAS score for back and leg pain (Figure 3). When comparing the two groups, there was no significant difference in the improvement of leg pain (6.57±0.78 vs. 6.13±0.64, p=0.29; Figure 4A). However, the improvement in back pain was significantly higher in the PELD group than in the OLM group (6.42±1.13 vs. 4.38±0.91, p=0.05; Figure 4B).
In the PELD and OLM groups, all 15 patients showed an improvement in preoperative CES symptoms including impaired lower limb motor power, perineal sensations, anal sphincter tone and bladder function at the one-year follow-up.
The operation time (45.00±4.08 vs. 96.25±10.60, p=0.01) and length of hospital stay (2.43±0.53 vs. 10.63±2.26, p=0.01) were shorter in the PELD group compared with the OLM group. In the PELD group, the intraoperative bleeding was negligible whereas in the OLM group, the estimated bleeding was 235±105 mL.
No patients suffered from disc recurrence, postoperative infection, or segmental instability requiring fusion surgery during postoperative follow-up.
DISCUSSION
CES is a very rare condition. That accounts for 1% to 3% of all lumbar disc herniations [29-31]. CES is usually characterized by so-called ‘red flag’ symptoms including severe low back pain, sciatica (often bilateral but sometimes absent, especially at L5/S1 with inferior sequestration), saddle and/or genital sensory disturbance, and bladder, bowel, and sexual dysfunction.
CES can seriously impair the quality of life without adequate treatment. Busse et al. [32] reported a strong correlation between long CES symptom duration and poor functional outcome. Beyond 24 hours, decompression delay may be associated with a poorer quality of life but, because of the rarity of CES, the sample size in this study was too small to provide definitive conclusions.
Lam et al. [33] showed the long-term prevalence of CES related bladder, bowel, and sexual dysfunction and their impact on the quality of life to inform service provisions. Overall, 71 patients (42 males, 29 females) were enrolled. When post-CES was compared with pre-CES, there was a higher prevalence and significant intrarespondent deterioration of bowel dysfunction, bladder dysfunction, perception of bladder function, sexual function, effect of back pain on sex life and activities of daily living/quality of life (p<0.0001 for all). Significant differences in individual questions asked pre-CES versus post-CES were also found.
The aim of our study was to examine differences in clinical outcomes between PELD and OLM. The result of our study showed that there were no significant differences between the two procedures.
PELD has several disadvantages regarding its limited field of view including difficult bleeding control, and because of the working channel, limited instruments can be used during the procedure.
Especially, increased intradiscal pressure resulting from insertion in the working channel, results in nerve root compression. However, in the case of degenerative disc disease, several studies have reported that the intradiscal pressure was low.
Sato et al. [34] measured the intradiscal pressure (vertical and horizontal) using an advanced pressure sensor in 8 healthy volunteers and 28 patients with ongoing low back pain, sciatica, or both at L4/5. They concluded that the intradiscal pressure in degenerated discs was significantly reduced compared with that of normal discs.
Schnake et al. [35] showed that at the beginning of the degenerative course, the water content of the nucleus pulposus was decreased and the proteoglycane composition was altered. This led to reduction of the intradiscal pressure.
In contrast, performing OLM requires the retraction of the nerve root. In the case of CES, nerve root retraction result in worse neurologic outcomes.
Several studies have reported the advantages of PELD, and our study confirmed these advantages including less soft tissue trauma, better bony preservation, rapid recovery, and avoiding root retraction.
On the basis of these advantages, several studies have demonstrated the better outcomes of CES treated by PELD [26-28].
Chen et al. [26] studied 11 cases of CES caused by lumbar disc herniation. After emergent surgery with PELD, the lower extremity symptoms were completely recovered or partly decreased. The decreased perianal sensations were partly recovered after surgery, and 9 cases had complete recovery and 2 cases had partial recovery at the one-year follow-up. No patients had anal contraction or bladder problems after the one-year follow-up.
Krishnan et al. [27] reviewed 15 patients who underwent percutaneous transforaminal endoscopic lumbar discectomy (PTELD) under local anesthesia. Ten patients underwent CESI and five patients received CESR. Bladder symptom recovery was 100%, and motor recovery was 80%. The VAS for back pain recovered to 0.53 from 8.00 and the VAS for leg pain recovered to 0.13 from 9.20. The ODI improved to 6.07 from 77.52 and the time to recovery bladder function was 1.47 days. Abnormal PVR urine was normalized in CESR patients at five weeks post-operation.
Li et al. [28] reported the results of 16 CES patients treated by PELD. There was a significant difference in the VAS for leg and back pain between preoperative and 1 day postoperative (p=0.007, p=0.01) as well as between preoperative and last follow-up (p=0.007, p=0.003). Three patients had residual saddle anesthesia remaining at last follow-up; however, these three patients’ preoperative radicular pain was relieved. Based on the Macnab criteria, the outcomes were excellent in 7 of 16 patients (43.8%), good in 6 patients (37.5%), and fair in 3 patients (18.7%).
CONCLUSION
In conclusion, there were no significant differences between PELD and OLM in clinical outcomes for CES. The advantages of PELD (the procedure can be performed under local anesthesia, less soft tissue trauma, better bony preservation, rapid recovery, and avoiding root retraction), indicate it is a good alternative or option for the treatment of CES patients considering the appropriate indication.