Lateral Lumbar Interbody Fusion in the Outpatient Setting with Multimodal Analgesic Protocol: Clinical Case Series
Article information
Abstract
Objective
Minimally invasive techniques and multimodal analgesia protocols have made spine surgery in the outpatient setting increasingly feasible. A number of spinal procedures have been documented in the outpatient setting, though the feasibility of lateral lumbar interbody fusion (LLIF) on an ambulatory basis has not been thoroughly assessed. To present a clinical case series of patients undergoing LLIF in the outpatient setting.
Methods
A prospectively maintained surgical database was retrospectively reviewed to identify patients undergoing outpatient spine procedures with an enhanced multimodal analgesia protocol from October 2016 to February 2021. Patient demographics, medical and spinal diagnoses, procedural characteristics, operative duration, estimated blood loss (EBL), postoperative length of stay (LOS), postoperative pain scores, postoperative narcotic consumption, and incidence of any intra- or postoperative complications were collected. The state’s prescription monitoring program was queried to assess rates of filling narcotic prescriptions >6 weeks following surgery.
Results
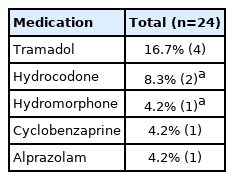
A total of 24 LLIF patients were included. Mean postoperative pain score was 5.8, and mean postoperative narcotic consumption was 26.8 oral morphine equivalents. All patients were discharged on the same day of surgery. No postoperative complications were observed. After the 6-week postoperative timepoint, 16.7% of patients filled a prescription for tramadol, 8.3% for hydrocodone, 4.2% for hydromorphone, 4.2% for cyclobenzaprine, and 4.2% for alprazolam.
Conclusion
This clinical case series demonstrates that LLIF can be both safe and feasible in the outpatient setting, with minimal narcotic medication dependence in the postoperative period.
INTRODUCTION
Introduced by Pimenta et al. in 2006 as an alternative to anterior or posterior approaches for lumbar arthrodesis, the retroperitoneal transpsoas minimally invasive lateral interbody fusion (MIS LIF) or lateral lumbar interbody fusions (LLIF) provides an effective approach for surgical interbody fusion for a broad spectrum of surgical pathology [1,2]. The LLIF has become increasingly adopted by spine surgeons due to its beneficial approach (avoiding spinal canal and neural foramen dissection), biomechanics (wide interbody cage spanning the dense apophyseal ring), and minimally invasive technique. In this technique, the spine is accessed laterally via splitting fibers of the psoas muscle longitudinally to create a retroperitoneal corridor [3]. The lateral plane of access affords the surgeon wide discectomy similar to an anterior approach, with the benefit of avoiding major bowel and vessel injury anteriorly and nerve and dural damage posteriorly [4]. A testament to its utility, the indications for the technique have broadened since its inception, and recently it has started being performed in the outpatient setting [4].
With the advancement of minimally invasive spinal operative techniques coinciding with milestone developments in anesthetics along with post-operative pain management protocols, spinal fusion surgeries are becoming increasingly feasible within outpatient ambulatory settings with several studies documenting its safety, efficacy, and beneficial cost profile when compared to inpatient surgery [4-8]. Further, the renewed emphasis of value and cost containment in spine surgery has served as a further impetus to the transition of surgical cases traditionally performed in an inpatient setting to being performed increasingly in ambulatory surgery centers (ASC) [8]. Owing to the expanding role of outpatient spine surgery and inevitable growth of ASCs, understanding determinants and deterrents of outpatient spine surgical success is paramount to both surgeons and administrators.
A significant barrier in transitioning fusion surgery to the outpatient setting is optimizing postoperative patient pain, as prior studies have noted inadequate pain control as the leading factor underpinning unplanned postoperative admission [9]. While providers are afforded the flexibility to alter discharge plans in an inpatient setting, this is not feasible in the context of stand-alone ASCs. It is therefore crucial that a safe, effective, and reproducible multimodal analgesia (MMA) protocol is used in this setting. While studies have commented on such protocols for lumbar decompression, transforaminal lumbar interbody fusion, there is scarce literature documenting MMA protocols for LLIFs in an ambulatory setting [10,11].
To address this need, our study presents a clinical case series of patients undergoing LLIF with an enhanced multimodal analgesic protocol in an ambulatory setting. The goal of the protocol was to provide reproducible postoperative pain relief while reducing patient reliance on opioid medication. We believe our experience may be of use to surgical teams interested in adapting lateral fusion cases to an outpatient setting.
METHODS
1. Selection
After institutional review board approval (ORA #14051301) and patient-informed consent were obtained, a retrospective review for eligible patients who underwent LLIF between October 2016 and February 2021 was performed. Inclusion criteria consisted of primary or revision, single or multi-level LLIF procedures for degenerative pathology utilizing our MMA protocol (Appendix). Procedures indicated due to trauma, infection, or malignancy were excluded. All surgeries were performed by senior author who is fellowship trained attending spine surgeon, and all surgeries were conducted at ambulatory surgical center where observation >23 hours is not permitted.
2. Data Collection
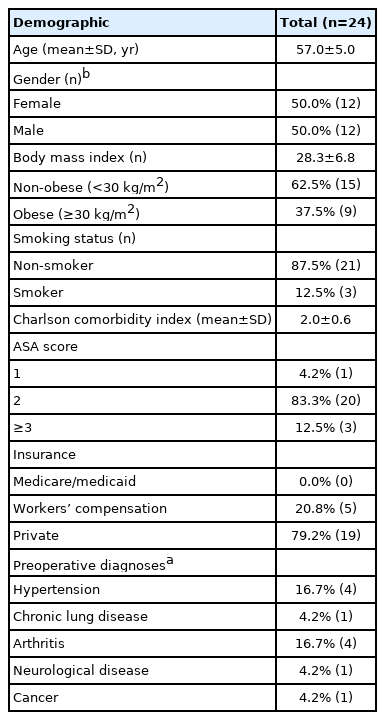
Selected baseline characteristics were noted, including age, sex, weight, BMI, comorbidity burden as determined by Charlson Comorbidity Index, smoking status, American Society of Anesthesiologists (ASA) score, and pre-operative spinal diagnosis (Table 1).
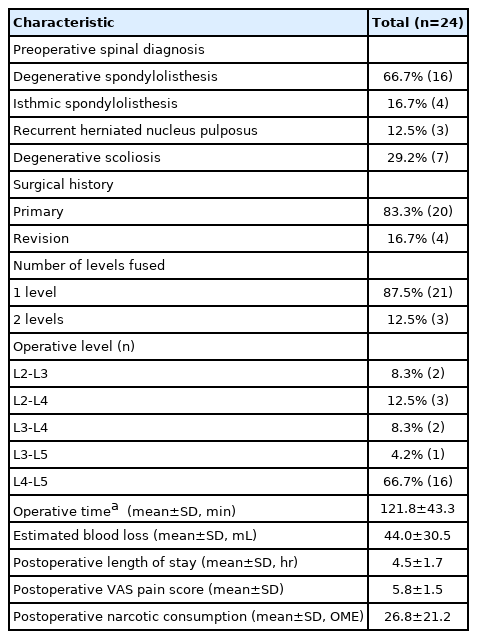
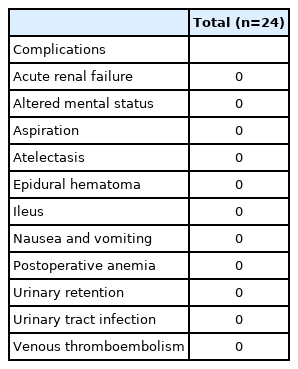
A range of intraoperative data was collected, including the primary or revision status, the number of operative levels, the index level, the operative duration (from the skin incision to closure), and blood loss estimates. Postoperative variables were recorded, including surgery center length of stay, patient-reported visual analog scale pain scores before discharge, and quantity of narcotic medications administered before discharge (i.e., converted into units of oral morphine equivalents and summed across all types of narcotic medications prescribed) (Table 2). Complications experienced during the immediate postoperative time period were recorded (Table 3). The state’s prescription monitoring program was queried to assess rates of filling narcotic prescriptions >6 weeks following surgery (Table 4).
1) MMA Protocol
A standardized protocol is initiated for all procedures and modified on an individualized basis. Successful administration of anesthetic regimen is initiated at patient’s preoperative visit, when he or she is informed of the perioperative procedure and expectations. Pre-emptive analgesia is administered before the start of surgery, consisting of cyclobenzaprine, pregabalin, and oxycodone. Intraoperatively, patients are induced using propofol and ketamine. Maintenance anesthesia consists of sevoflurane gas and a fentanyl infusion. Additional intraoperative medications include bupivacaine with epinephrine, acetaminophen, dexamethasone, ondansetron, and famotidine. Postoperatively, patients receive a detailed protocol of PO medications. Protocol can be viewed in Figure 1 and 2. The MMA protocol was indicated in patients undergoing LLIF in the outpatient ambulatory setting.
2) Surgical Technique
LLIF patients were placed in the lateral decubitus position and approach was transpsoas. The surgical procedure differed based on surgeon preference and patient needs, and consequently surgeries included LLIF with posterior fixation (rods and percutaneous screws) as well as stand-alone LLIF. The surgeries were all performed under neuromonitoring guidance. A single, transverse incision was made on the lateral aspect of the body in-line. The psoas muscle is palpated and blunt dissection is carried out through the psoas down to the disc space. The cannulated dilator was attached to the disc center, and a guide wire was fed through it. Before removal, a specialized retractor was placed over the last dilator. Muscle was cleared from the annulus, which was then incised. The contralateral annulus was perforated after discectomy and endplate preparation. Interbody cages were filled with allograft and then implanted. The patient was then moved from lateral decubitus to the prone position in cases requiring direct decompression. Finally, posterior decompression and bilateral pedicle screw placement were achieved via an 18-mm tubular retractor and guide wire, respectively.
RESULTS
1. Patient Demographics
Total of 24 LLIF patients were included with a mean age of 57.0 years, of whom 50.0% were female and 37.5% were obese. Hypertension was the most common preoperative diagnosis and majority (79.2%) of patients utilized private insurance. Mean Charlson Comorbidity Index was 2.0 and most patients had an American Society of Anesthesiologists classification of 2 (83.3%) (Table 1). Most patients had a preoperative spinal diagnosis of degenerative spondylolisthesis (66.7%) and underwent primary (81.8%) single-level (87.5%) procedures.
2. Perioperative and Postoperative Outcomes
Majority (83.3%) of surgeries undertaken were primary. The most common level to be fused was L4-L5 (66.7%), and most cases were single-level surgeries (87.5%). Mean operative duration was 121.8 minutes, mean EBL was 44.0 mL, mean LOS was 4.5 hours, mean postoperative pain score was 5.8, and mean postoperative narcotic consumption was 26.8 oral morphine equivalents (Table 2). All patients were discharged on the same day of surgery. No postoperative complications were observed (Table 3). After the 6-week postoperative timepoint, 16.7% of patients filled a prescription for tramadol, 8.3% for hydrocodone, 4.2% for hydromorphone, 4.2% for cyclobenzaprine, and 4.2% for alprazolam (Table 4).
DISCUSSION
In the United States, surgical care accounts for 7% of the gross domestic product (GDP) [12]. As healthcare costs continue to increase, hospitals, patients, and insurance providers are actively working to reduce costs and patients’ length of stay without compromising the quality of care and improving the value of care. As advancements in operative, analgesic, and anesthetic techniques improve, surgical treatment in outpatient ambulatory surgery centers (ASCs) appears to be more favorable than inpatient hospital settings. While implementation of such advances present potential clinical and economic gains, thorough evaluation of procedure efficacy and patient outcomes is required as the quality of care and patient safety are not to be compromised.
In this study, we present a case series of 24 patients undergoing LLIF surgical treatment in an outpatient setting utilizing our institution’s novel MMA protocol. While studies have commented on similar analgesic protocols for lumbar decompression, transforaminal lumbar interbody fusion, there is scarce literature documenting analogous protocols for the lateral approach to fusion in an ambulatory setting [10,11]. Additionally, it should be noted that the protocol utilized in this surgery for LLIF technique is not specific to the lateral approach but may be generalized per surgeon discretion to other fusion surgeries performed in an ambulatory setting.
On average, patients were younger than 65 years old and had an average length of stay of 4.5 hours. Additionally, the mean narcotic consumption of postoperative day 0 (POD 0) was 26.8 oral morphine equivalents (OME). All patients did well during the surgery with none presenting any complications postoperatively and all patients were discharged home on the same day of surgery (in less than 23 hours) without further hospitalization or readmission required.
These positive results are attributed to our strict adherence to effective MMA protocol, appropriate pre-and peri-operative patient management, and effective patient selection. Prior studies have demonstrated MMA protocols following spine surgery to be instrumental in postoperative clinical improvements in pain and disability without raising patient complication profile [13,14].
1. Patient Selection
In order for successful surgery avoidant of potential complications, patients must be carefully selected for both the outpatient setting and for use under the MMA protocol. In their study, Smith et al. [15] concluded that the best predictors for early postoperative discharge and prevention of transfers to inpatient facilities from outpatient settings were decreased comorbidity burden, age, and BMI. Further, Chin et al. [16] proposed the following criteria for outpatient spine surgery based on their experience with lumbar decompression, disc replacement, and fusion in a single surgeon private practice: patients living or staying 30 minutes or less of the surgery location, a BMI≤42 kg/m2, clearance from a patient’s general practitioner or cardiologist, ASA score of ≤3, and presence of a responsible adult for at least 24 hours after the surgery. Using this criteria, the authors concluded that 79% of spine surgery patients in their private practice were eligible for procedures in the ambulatory setting [16].
The results of our study align with the findings of prior literature in that the majority of patients deemed suitable for outpatient LLIF were younger than 65 years old, had a BMI less than 42 kg/m2 and had an ASA score of less than 3.
Specific criteria for LLIF in ASCs have not yet been developed to our knowledge. In combination with published literature and our patient demographics in this study we present the following exclusion criteria: patients≥65 years older [12], BMI≥40 kg/m2 [16,17], increase risk of postoperative nausea and vomiting (PONV) [5,18], no presence of a responsible adult for at least 24 hours postoperatively or the patient cannot take care of him/herself [5,16], and an ASA score no greater than 3 [19]. In addition to these criteria, surgery in an ASC may not be possible if cases are composed of complex spinal pathologies and/or multilevel cases, more invasive procedures such as open approaches in place of MIS, and operations that must be done in parts or have duration greater than 2 hours [5,20].
Furthermore, ASCs may have limited available clinical support staff and emergency services. In case of an emergency, patients may be transferred to the hospital. As such, it is vital to acknowledge a patient’s comorbidities and health. In our study, no patients with an ASA greater than 3 were included. In this way, patients that have present comorbidities that may pose life-threatening risks are not included as the operative risk is too high.
2. Multimodal Analgesia Protocol
Our team’s MMA protocol aims to target multiple causes of pain following spine surgery, including neuropathic pain, inflammation, muscle spasticity, and a diminished central nervous system pain threshold [21]. With these aforementioned aims in mind, the protocol strives to optimize pain control and therefore reduce dependence on opioid medications [21]. Adequately controlling pain postoperatively has shown success in lowering risk of complications, faster recovery, superior mobility and coordination, and even patient satisfaction [22-24]. In a randomized controlled-trial by Kim et al. [14] of 80 patients undergoing lumbar spinal fusions, the authors reported that pain scores decreased and disability scores improved postoperatively without increasing complication rates. Further, in a retrospective review of postoperative clinical and perioperative outcomes following single-level transforaminal lumbar interbody fusion (TLIF) using either a MMA or patient-controlled analgesia (PCA) protocol, authors reported the MMA cohort to have reduced rates of inpatient narcotic consumption, PONV, and reduced LOS [17]. The same study noted, however, that postoperative narcotic consumption after discharge, inpatient pain scores, and Post operative urinary retention demonstrated no differences between the two cohorts [17].
In our study, the mean postoperative pain score was 5.8 on a numerical Visual Analog (VAS) scale of 0–10 (0 being no pain and 10 being as bad as it can be) (Table 2). Our team attributes our postoperative pain management primarily to the preoperative and intraoperative components of our analgesic protocol. Following orthopedic surgical procedures where severe pain can be experienced by the patient before the surgery and afterward, detailed preoperative analgesia is crucial to enhancing patient recovery, pain ratings, and rehabilitation [25,26].
3. Preoperative Component
Several approaches have been proposed to alleviate severe postoperative pain, such as use of preemptive analgesia, opioids, cyclooxygenase-2 inhibitors, epidural anesthesia, peripheral nerve blockade, local infiltration analgesia, patient-controlled analgesia, and multimodal analgesia [26]. Furthermore, Kashefi et al. [27] found preoperative administration of either tramadol, cyclooxygenase-2 inhibitors, nonsteroidal anti-inflammatory drugs or opioids lead to deceased patient-reported levels of pain, increased activity levels, improved mental health reporting, and generally a positive satisfaction with their procedure. Acetaminophen demonstrates the ability to enhance postoperative pain scores and analgesia predominantly through central nervous system action [28,29]. Beyond the well documented analgesic effects, acetaminophen also has antipyretic effects preventing downstream peroxidase steps involved with synthesis of prostaglandins [30]. Acetaminophen is inexpensive, is associated with few mild adverse side effects, and has demonstrated effectiveness, often as a single agent, in managing the analgesic effectiveness of surgery. Preoperative utilization of anticonvulsants, such as gabapentin and pregabalin, have demonstrated efficacy in reducing pain intensity postoperatively and improving functional outcomes for spine surgeries [31]. Turan et al. [32] specifically note in their study in which patients received 1,200 mg gabapentin 1 hour before surgery that patients reported decreased pain scores and decreased postoperative morphine use versus the placebo group following spine surgery. Further, the use of cyclobenzaprine preoperatively, often in conjunction with nonsteroidal anti-inflammatory drugs, have improved efficacy over opioids. Cyclobenzaprine has been observed to provide significant lower back pain relief justifying their utility as a multimodal analgesic approach [33].
4. Intraoperative Component
While the utility of preoperative pain management is gaining statistical and clinical significance, intraoperative pain management has long been a standard surgical approach. Medications that decrease the patient’s level of consciousness, along with inhaled anesthetics, are used to keep the patient comfortable during the procedure. Specifically propofol and ketamine [30]. Ketamine, which antagonizes an NMDA receptor, has been proven to decrease opioid consumption via receptor modulation and lowering central excitability also function in conjunction with acetaminophen and nonsteroidal anti-inflammatory drugs intraoperatively. Throughout the surgical procedure, acetaminophen, dexamethasone, ondansetron, and famotidine are given [25,30]. Further, intraoperative use of bupivacaine was administered in a one-time dose typically towards the conclusion of surgery to provide significant incremental pain relief. A study by Reynolds et al. [34] demonstrated 0.25% solution of bupivacaine associated with decreased opioid use following fusion surgery for idiopathic scoliosis compared to patients receiving local anesthetic, without showing systemic side effects.
5. Postoperative Component
By providing adequate postoperative analgesia, patients would not only experience less pain and possibly less opioid consumption, but it also has implications for cost savings and improved rehabilitation by reducing the length of stay, conducting more effective postoperative therapy, and increasing patient satisfaction [26,35]. MMA protocols optimize non-opioid interventions by limiting their use to treat “breakthrough” pain postoperatively. Furthermore, the mean postoperative narcotic consumption on the day of surgery was 26.8 oral morphine equivalents (OME). After querying the state’s prescription monitoring program to assess rates of fulfilled narcotic prescriptions at 6 weeks following surgery, it was shown that 16.7% of patients filled a prescription for tramadol, 8.3% for hydrocodone, 4.2% for hydromorphone, 4.2% for cyclobenzaprine, and 4.2% for alprazolam demonstrating that the majority of patients had weaned off narcotics. It should be noted that regarding opioid utilization following MMA protocol in this case series, at the 6-week timepoint 4 patients utilized tramadol, 2 patients utilized hydrocodone, 1 patient utilized hydromorphone, 1 patient utilized cyclobenzaprine, and 1 patient utilized alprazolam. 1 patient received both medications at different timepoints. Regarding opioid medication usage at 6-weeks, 5 patients or around 20% of patients in this case series utilized a narcotics at 6-weeks. When combined with opioid equivalent analgesics, about 1/3 of case series fulfilled medication prescription at 6-week. This would suggest that a non-trivial number (20%) of patients required narcotic medications at 6-weeks, and further study with greater cohort sizes will need to be conducted to understand how to drive this percentage lower. Although this case series consists of only 24 patients, the results of our study as a preliminary analysis show the promise and utility of our MMA protocol in the context of outpatient LLIF surgery as the majority of patients in this case series were weaned off narcotic medications at first follow-up. The overarching goals of an MMA protocol include using a variety of medications to reduce perioperative pain, specifically improving patient-reported postoperative pain ratings while minimizing harmful iatrogenic effects. Postoperatively, patients are given a detailed outline of PO medications. Many preoperative medications, including muscle relaxants, anticonvulsants, and opioids, are continued into the postoperative period. Additionally, a schedule including NSAIDs and/or acetaminophen is part of a comprehensive MMA [30]. While surgical interventions may be the next best step in treating a pathological state, it doesn’t come without risk of pain leading to stress, anxiety, depression, and medication dependence. The development and implementation of a thorough patient-centered MMA can decrease postoperative pain-related risks and lower dependence on opioids long term. Implementation and utilization of a patient-centered MMA can adequately reduce pain and improve patient-reported outcomes.
6. Complications
Unique to the LLIF, it involves exposure to the lumbar spine through a lateral aspect by a retroperitoneal approach through the psoas muscle [36]. For successful completion of an LLIF procedure, preoperative imaging in the form of MRI or CT is necessary to visualize the disc space and ensure that intra-abdominal vessels are not vulnerable to injury [36]. Throughout the procedure, neuromonitoring and fluoroscopy are utilized to obtain anterior-posterior and lateral radiographs for the surgeon to plan and perfectly execute the incision in line with the disc space [36].
Reported complication rates are controversial across literature [37], which most likely is associated with the surgeon’s surgical experience, patient demographic characteristics, and varied instruments and/or techniques utilized [38]. The most common and greatest risk from LLIF includes neurological injury to the lumbar plexus, which may result in sensory and motor problems potentially due to psoas muscle trauma or lumbar plexus stretching. It should be noted that an anterior to psoas approach not utilized in this case series may provide a more natural corridor to the disc space with decreased postoperative risk of hip weakness with such risk inherently greater in the lateral transpoas approach [39].
As the lateral approach to fusion is technically demanding and post-operative complications are more likely the longer a procedure lasts, surgeons unable to perform cases in <2 hours should not migrate lateral fusion cases to outpatient setting. In a multicenter study of prospectively treated patients fused at L3-4 and L4-5 with LLIF, 27.5% of patients reported postoperative hip flexion weakness and the systematic review by Hijji et al. [37] noted that up to 36% of patients reported temporary neurologic injury following surgery with lumbar neurapraxia occurring in up to 62.7% of patients [40]. Previous studies have attributed such complications to prolonged retractor time (>30 minutes), increased tissue edema and damage from retractor placement, and excess extension of the psoas muscle opening leading to ischemic nerve damage [37,40]. Performing an open LLIF is especially not suitable, as these procedures are generally longer and patients require longer time to recover, often requiring hospitalization [41]. In our study, the attending surgeon benefitted from 15 years of experience with MIS techniques, allowing for shortened operative times, minimized blood loss, and tissue trauma burden. Surgical experience and where a surgeon is on their learning curve is important when assessing when to make the switch from performing lateral fusions in an inpatient setting to the ASC.
Rarely, bowel perforation has been reported as a life threatening complication of the lateral approach, with literature noting that primary means of avoidance of bowel perforation to be presurgical imaging (MRI/CT) to provide information regarding sagittal vertebral length and anatomical abnormalities [2,42]. Furthermore, by examining the location of anatomic structures, such as the retroperitoneal vasculature, lumbar nerve roots, and genitofemoral nerve relative to the psoas muscle, complications related to retroperitoneal exposure may be reduced [42].
Postoperatively, the lateral approach has been associated with ileus, with independent risk factors including GERD, L1-2 operative levels, and posterior instrumentation [43]. A multimodal approach should be utilized to limit administration of narcotics and other agents that may contribute to decreased gastrointestinal motility [44].
In our study, no immediate postoperative complications were observed (no cases of acute renal failure, altered mental status, aspiration, atelectasis, Epidural hematoma, ileus, nausea and vomiting, anemia, urinary retention, urinary tract infection, or venous thromboembolism) (Table 3). This is surely attributable to surgeon experience, low study power, and patient selection. As careful attention was paid to selection of patients for outpatient spine surgery, this allowed for faster recovery and discharge from the ASCs. Patients included in this outpatient case series were generally younger (mean age of 57.0 years), majority were non-obese (62.5%), with low independent comorbidity burden (83.2% with ASA score<3). These modifiers of patient risk surely benefited our postoperative complication results as elevated comorbidity, age and BMI are risk factors for surgical complication postoperatively. As such conclusions of the utility of this protocol and subsequent risk of postoperative complications in older patients, obese patients, or those with severe comorbidity burden should not be made. Furthermore, a significant barrier to discharge is postoperative nausea and vomiting, which notably in our case series no patients were observed to display. While the small sample size could have played a role, the reliance of our protocol on local anesthesia that doesn’t exert systemic effects potentially contributed to this observation. In our protocol, appropriate treatment for PONV includes preoperative administration of anti-emetics such as ondansetron or metoclopramide, and adequate hydration.
7. Limitations
As our study is a case series, only 24 patients were included which limits the power of the study. Additionally, not all of the patients the surgeon treated were included in the study due to refusal to consent to the study on the patient’s behalf. Further, we did not follow up with the patients or analyze patient-reported outcomes, which if included would add strength to the study with insight into patient postoperative improvement across quality of life domains. Given strict selection for outpatient ambulatory spine surgery, patients included in this case series were generally younger, non-obese, with low comorbidity burden. As such conclusions of the utility of this protocol in older patients, obese patients, or those with severe comorbidity burden should not be made. Additionally, this study is based on a single surgeon at a single academic institution limiting generalizability of study findings to other clinical settings. In addition, while mention of opioid prescriptions more than 6 weeks postoperatively is reported, we did not report whether some of these patients were opioid-naive patients or patients already taking opioids.
CONCLUSION
Ambulatory surgery centers are performing a growing number of spine surgeries. With the proven benefits and improving safety profile of the lateral approach, an increasing number of LLIFs will be performed in ASCs. The ability to control pain is a crucial component of the success of these procedures. Based on a review of high-quality literature and our own clinical experience, we present a specific multimodal protocol in this study. The findings of our study may inspire similar groups to adopt aspects of their own MMA protocols.
Notes
No potential conflict of interest relevant to this article.