AbstractObjectiveThe aim of this study was to assess whether robotic assisted surgery is more efficient, in terms of estimated blood loss (EBL), operating time, post-operative pain scores, hospital stay and discharge disposition, than standard navigated MIS techniques when performing a transforaminal lumbar interbody fusion (TLIF).
MethodsRetrospective chart review was performed for 1,027 consecutive patients for a single spine surgeon. One-hundred patients who underwent either navigated MIS TLIF or robotic assisted MIS TLIF procedures were identified and included in the study. Primary outcome measures included EBL, operating time, post-operative pain scores, length of hospital stay and discharge disposition. Baseline demographic data, as well as outcome data, was recorded for each patient. Statistical analysis was performed to determine differences between the two groups.
ResultsPatients who underwent robotic assisted MIS TLIF surgery had less intra-operative EBL (p=0.002), shorter operation time (p<0.001) and a shorter hospital stay (p=0.036) than those who underwent navigated MIS TLIF surgery without robotic assistance. There was no difference in discharge disposition or post-operative pain scores between the groups.
INTRODUCTIONSpine surgery is among the leading specialties in terms of technological advancements in surgery. There is a constant search for novel techniques and tools that allow for better patient experiences and improved outcomes. The use of minimally invasive surgery (MIS) techniques instead of traditional open approaches has been one of these major advancements [1]. Previous research has indicated that the MIS Transforaminal Lumbar Interbody Fusion (TLIF) technique results in less operative blood loss, less post-operative pain, shorter hospital stays, and decreased narcotic use compared to the open TLIF technique [2-4]. This is thought to be the result of decreased muscle stripping and iatrogenic soft tissue injury. However, long term clinical outcomes have been found to be similar between both techniques, with both producing excellent results [3].
As the field of spine surgery continues to evolve, the technology and techniques aim to minimize the burden of recovery while simultaneously maximizing clinical benefits. The next significant advancement after MIS was the use of intra-operative navigation software. This allowed surgeons to more accurately place pedicle screws compared to the use of intra-operative fluoroscopy [5]. To further improve accuracy and efficiency, robotic assistance has been proposed. In theory, robotic assistance can make hardware placement easier for surgeons, resulting in less collateral damage to the soft tissues. The robotic arm is not only used to place hardware, but also to align skin and fascial incisions at the exact angle and orientation needed for screw placement. This allows for minimal soft tissue dissection.
Previous studies have suggested that the soft tissue injury sustained during open surgery is responsible for the increased blood loss, pain, recovery time and hospital stay associated with the technique [6]. This principle is the basis for the improved surgical parameters associated with MIS TLIF surgery over open TLIF surgery. Navigation software further decreases the need for extensive dissection, but most techniques recommend an abbreviated approach to the screw entry point for ideal hardware placement. In contrast, robotic assisted MIS techniques suggest percutaneous, sharp incision of the skin and fascia with a scalpel as the only dissection for the approach. Performing this through the robotic arm provides a direct path for screw placement without further dissection or exposure of the starting point. Theoretically, this lack of dissection would result in even less soft tissue damage, and thus patients might experience an even more comfortable and accelerated recovery course.
The use of a robot for pedicle screw placement also has several other possible benefits. These include reliable and reproduceable placement of pedicle screws, resulting in a more standardized screw placement process. This could lead to more consistent outcomes across different patients, as well as for the development of more comparable research studies. It may also improve results and decrease complications for less experienced surgeons, especially early in their careers. Another benefit of robotic assistance is that surgeons can account for variations in patient anatomy, from complex deformities to occult dysmorphisms [7]. This more accurate anatomic placement of hardware could result in biomechanical advantages over free hand placement [8]. Using a robot may also allow for quicker pedicle screw placement, as there are less variables during the placement process. This would significantly shorten operating time, depending on the number of screws being placed. Finally, the use of a robot for hardware placement may allow increased resident and trainee participation in screw placement, as attending surgeons can easily visualize the location of the screw on the monitor, and it is relatively difficult to greatly deviate from the planned insertion location.
While the traditional MIS TLIF technique has been frequently compared to open surgery, there is little data comparing the navigated MIS TLIF procedure to robotic assisted MIS TLIF surgery. This study aims to evaluate whether robotic assisted pedicle screw placement during navigated MIS TLIF surgery further optimizes EBL, OR time, post-operative pain scores, hospital stays, and discharge disposition.
MATERIALS AND METHODS1. Study Design and Patient SampleAfter Institutional Review Board approval was obtained, this retrospective chart review included patients who underwent surgery by a single fellowship-trained spine surgeon between May 2017 and April 2020. All surgeries were performed at a single institution. Patients that required additional procedures, such as a laminectomy, were excluded. Other exclusion criteria included patients undergoing revision surgery, constructs that included thoracic levels, and those that included more than two surgical levels. There were no exclusions based on age, comorbidities, or other demographic information.
2. Data CollectionBaseline patient data collection included patient sex, age, body mass index (BMI), American Society of Anesthesiologists (ASA) status, and pre-operative diagnosis. The type of surgery performed, estimated blood loss, operating room time, discharge disposition and daily pain scores were also collected from the patients’ electronic health records (EHR). Operating time was calculated from the time of incision until the time nursing staff marked the end of procedure, usually shortly after skin closure. Length of stay was calculated in hours from the time the patient left the operating room until they were discharged. All the collected data was then compared between the Robotic Assisted and the standard navigated MIS surgical groups.
3. Navigated MIS TLIF Surgical TechniqueThe incision was performed in a stab wound fashion over the left posterior superior iliac spine with percutaneous placement of a Medtronic (Minneapolis, MN) Stealth reference for the navigation. Intraoperative imaging of the lumbar spine was obtained using the Medtronic O-arm navigation system. Stereotactic navigation was used for skin marking for entry into the designated pedicles bilaterally. Incisions with a #10 scalpel were made and subdermal dissection performed with monopolar cautery. Fascial incision and subfascial dissection were performed with cautery until the desired screw starting point was identified. Under stereotactic guidance, insertion of pedicle screws was performed with the Stryker (Kalamazoo, MI) ES2 system. A pedicle base retractor was used on the side of more radicular symptoms to expose the facet complex. Intraoperative microscope was brought into the field. An en-bloc facetectomy was performed utilizing a high-speed burr, osteotomes and Kerrisons. The exiting and traversing nerve roots were then visualized and decompressed. The thecal sac was mobilized medially using a nerve root retractor over the shoulder of the traversing nerve root. A box-shaped annulotomy was performed with a #15 scalpel followed by thorough diskectomy using pituitary and Kerrison rongeurs. Paddle distractors were utilized, and endplate preparation was performed with shavers and rasp instruments. The field was copiously irrigated with antibiotic solution. Packing of the contralateral disk space with bone allograft and autograft was performed followed by insertion of a Stryker Titanium spacer, prepacked with bone allograft. The rods were inserted through the pedicle screws and appropriately fastened. Closure of the fascia was performed with 0 Vicryl interrupted, followed by closure of the subdermal tissue with 2-0 Vicryl interrupted. Closure of the skin as performed with 4-0 Monocryl in subcuticular fashion.
4. Robotic Assisted MIS TLIF Surgical TechniqueThe robotic assisted technique was performed in a similar fashion to the standard navigated MIS technique with the following differences. Instead of placing the Stryker E2 stealth navigation reference, the Globus Excelsius navigation reference was placed (Figure 1). The navigation system was then positioned, and intra-operative X Rays were taken. These images were then merged with the pre-operative CT scan. The robotic arm was used to mark out the location of pedicle screw incisions at the designated levels. A stab incision through skin, and subsequently fascia, was performed through the robotic arm with a 10-blade scalpel. The Globus CREO pedicle screws were inserted under robotic stereotactic guidance through the robotic arm (Figures 2, 3). Decompression was performed in the same fashion as previously described, with the only difference being that a Globus cage was used. The remainder of the procedure was performed identical to the navigated MIS TLIF technique described previously.
5. Statistical AnalysisUnivariate comparisons between patients in the robotic and non-robotic surgical groups were compared using independent t-tests for continuous variables and either chi-square tests of independence or Fisher exact tests for categorical variables. The alpha level was set at p=0.05 for all comparisons. A post-hoc power analysis using a two-tailed test with a moderate effect size (d=0.5) and an alpha level of p=0.05, showed the obtained power (1–b)=0.70. All analyses were conducted in SPSS version 24 (IBM, Inc.).
RESULTS1. Demographic and Diagnostic DataCharts of 1,027 adult patients were reviewed. Those who underwent Navigated MIS TLIF or Robotic Assisted MIS TLIF lumbar surgery were identified, resulting in 137 patient charts. The final number of patients included was 100, of which 52 underwent navigated MIS TLIF and 48 underwent Robotic Assisted MIS TLIF surgery. There were no statistically significant differences between baseline patient demographics. Mean age was 56.3 (SD 13.1) in the non-robotic group and 58.2 (SD 13.2) in the robotic group (p=0.491). The non-robotic group was made up of 34.6% males while the robotic group was 50% male (p=0.119). Average BMI in the non-robotic group 32.5 (SD 5.0) and was 33.0 (SD 6.8) in the robotic group (p=0.663). In the non-robotic group 31 patients underwent a single level operation and 21 underwent a two-level operation, while in the robotic group there were 31 single level operations and 17 two level operations (p=0.61). The primary diagnoses and ASA scores between the two groups were not statistically different (p=0.587 and p=0.812, respectively) (Table 1).
2. Surgical ComparisonsThere were several statistically significant findings between groups regarding surgical parameters (Figure 4). In the non-robotic group, mean EBL was 76.5 mL (SD 82.1), while in the robotic group mean EBL was 35.6 mL (SD 36.7) (p=.002). Average operating room time was 187.1 minutes (SD 52.3) for non-robotic surgery and 152.3 minutes (SD 40.6) for robotic-assisted surgery (p<0.001). Mean length of hospital stay was 92.3 hours (SD 50.1) in the non-robotic group and 71.6 hours (SD 47.1) in the robotic group (p=0.036). There was no significant difference in discharge disposition between the two groups (Table 2). There was also no significant difference in daily mean post-operative pain scores between the two groups for post-operative days 0–5 (Table 3).
DISCUSSIONThe goal of modern-day spine surgery is to provide maximum symptom relief to patients in the most efficient manner possible. Ideally, surgeons minimize blood loss, length of hospital stay, OR time, cost, post-operative pain, and narcotic use without sacrificing surgical outcomes. This principle has been consistently studied over time and is evident in the shift from open surgery to more MIS techniques [9]. The pathophysiologic explanation for MIS benefits is that by being more accurate with hardware placement and dissection techniques, patients undergo less collateral trauma during surgery. By inflicting less unnecessary trauma, patients experience less soft tissue damage, resulting in improved surgical parameters and outcomes [10]. This study aimed to further explore this idea by examining whether robotic assisted surgery in conjunction with navigated MIS techniques would improve the efficiency of TLIF surgery. We hypothesized that the robotic assisted technique would favorably affect surgical parameters in comparison to standard navigated MIS techniques alone.
It was found that patients who underwent robotic assisted MIS TLIF surgery experienced improved surgical parameters in all measured outcomes except post-operative pain scores and discharge disposition. The robotic assisted group had a surgical EBL of 35.6 mL compared to 76.5 mL in the non-robotic group (p=0.002). Average OR time was 152.3 minutes for the robotic assisted group and 187.1 minutes in the non-robotic group (p<0.001). Mean length of hospital stay was 71.6 hours in the robotic group while the non-robotic grouped averaged 92.3-hour hospital stays (p=0.036). There were no differences in post-operative pain scores or patient disposition between groups.
The difference in EBL between groups is likely related to the lack of dissection performed in the robotic-assisted group. The robotic arm alignment allows the surgeon to only incise the skin and fascia using a 10-blade scalpel, without further dissection, limiting soft tissue trauma. This results in less bleeding and thus less EBL. While the non-robotic group still utilized a navigated MIS technique, there was some cautery dissection performed. This is often necessary to acquire the correct starting point and trajectory for screw placement, as the navigation provides a general, but not perfect, location for the skin and fascial incisions. Similarly, the shorter hospital stay in the robotic-assisted group may also be related to a more minimal dissection. Less collateral soft tissue damage likely results in less post-operative pain. Less pain may result in improved ambulation and quicker discharge times. Unfortunately, the current study could not evaluate post-operative pain scores for reasons described later in this discussion.
To date, we are unaware of any other studies that specifically evaluate surgical parameters regarding the use of robotic assistance in MIS TLIF surgery compared to navigated MIS techniques alone. However, several studies have performed similar evaluations. A study by Cui et al. [11] compared traditional open techniques to robot-assisted MIS TLIF techniques in 41 patients with lumbar spondylolisthesis. They found that the robot-assisted group had less surgical EBL, shorter hospitalization, less post-operative pain and were quicker to ambulate. However, the robotic assisted group required longer operative time and more X-ray exposure.
The current study found similar results when comparing robotic-assisted MIS to standard navigated MIS TLIF surgery, except that the robotic group had shorter OR times. The shorter OR times for the robotic group in our study may result from several factors. First, the robotic-assisted surgeries did not involve use of the O-arm but did require merging of the pre-operative CT with the intra-operative X-rays. While the specific time required for these processes was not measured in this study, it is likely that the use of the O-arm in the non-robotic group took longer, contributing to a longer procedure time in that group. These processes were both accounted for in the overall OR time calculations since the start time was marked by the incision for the navigation reference, which was placed at the beginning of each procedure. It seems appropriate to account for these times as they do contribute to a patient’s time under anesthesia. Another factor that likely influenced the significant difference in OR times between groups is the speed of pedicle screw placement. When inserting pedicle screws using strictly navigation, the surgeon must rely on manual manipulation and orientation of the instruments to match the trajectory displayed on the navigation software. In contrast, when using robotic assistance, the surgeon simply places instruments through the robotic arm at the pre-determined angle. While the former technique may differ significantly based on surgeon experience, the latter is likely to be quicker and more consistent across different surgeons.
A recent study by De Biase et al. [12] focused on surgical outcomes between robotic-assisted TLIF and MIS TLIF patient cohorts. In this study, 101 patients of a single surgeon were utilized. It was found that there were no differences in post-operative complications or rates of surgical revision. This suggests that robotic assisted TLIF surgery is an effective alternative to MIS TLIF surgery alone in terms of patient outcomes. Interestingly, they did not find a difference in operative time between the groups. This may have been the result of surgeon technique, surgeon experience, or type of robot used, among other variables.
The current study has several limitations. First, all patients were treated by a single surgeon, limiting the generalizability of the findings. This surgeon also frequently had residents on his service. While the residents were involved in pedicle screw placement for patients in both groups, the presence and amount of involvement of these residents was not accounted for in this study. Future studies might include multiple surgeons and exclude cases where residents were present.
Data collection for pain scores and post-operative narcotic pain medication was difficult due to the nature of the electronic medical record (EMR) at the participating hospital. Pain scores were inconsistently documented and collected from a wide range of different nurses, likely resulting in high inter-observer variability. In terms of pain medications, the EMR did not easily allow for retroactive tabulation of pain medication doses. This information was altogether unavailable for some patients and thus was unable to be incorporated into the study. Unfortunately, this not only removed the ability to examine post-operative narcotic use, but also renders our post-operative pain scores relatively useless. Patients who were in more pain likely took more narcotic medication, bringing their pain level down. Other patients, who did not have as much pain, likely used less narcotics, ultimately resulting in similar pain scores for all patients. This phenomenon may have been the reason that we did not find any differences in post-operative pain scores between the two groups. Without accurate pain medication information, it is difficult to appropriately assess post-operative pain scores. Future studies should have a consistent and clear method for documenting pain scores and post-operative pain medications.
A future prospective study might include measuring the time taken for merging intra-operative X-rays with the pre-operative CT in the robotic assisted group, as well as the time used for O-arm operation in the non-robotic group. Time spent specifically on inserting pedicle screws would also be useful in determining ease-of-use differences between standard navigation and robotic-assistance. The accuracy of each method could then be evaluated utilizing post-operative CT scans. A concurrent cost analysis would be beneficial in comparing overall efficiency.
CONCLUSIONRobotic-assisted MIS TLIF surgery resulted in less EBL, shorter OR time and shorter hospital stays when compared to traditional navigated MIS TLIF surgery. Previous studies have found robot-assisted spine surgery to be effective in improving the accuracy of pedicle screw placement and decreasing radiation exposure for both the patient and surgeon when compared to traditional techniques [13-15]. Patient-oriented outcomes have also been found to be similar [16]. Few studies have elaborated on this concept and compared robotic-assisted MIS surgery to navigated MIS surgery, as we have done in this study. A series of prospective, randomized control trials that examine surgical parameters, as well as patient-oriented outcomes, would be ideal for further exploration into the benefits of robotic assisted spine surgery.
ACKNOWLEDGEMENTSThe authors are thankful to Krista Howard for assistance with statistical analysis.
Fig. 1.Globus Excelsius Robotic Navigation Platform setup. The navigation reference is visible in the patient’s right posterior superior iliac spine. Robotic arm is in position for left sided pedicle screw placement. 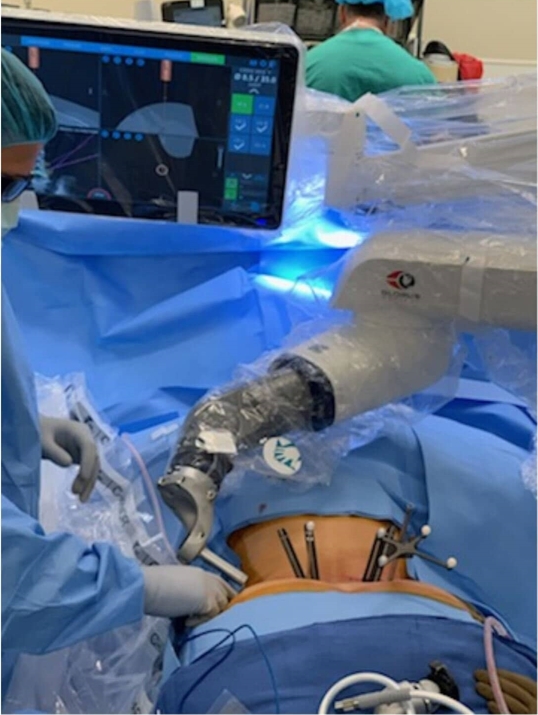
Fig. 2.Globus Excelsius Robotic Navigation software interface depicting the planned and real-time trajectory of the pedicle screw. 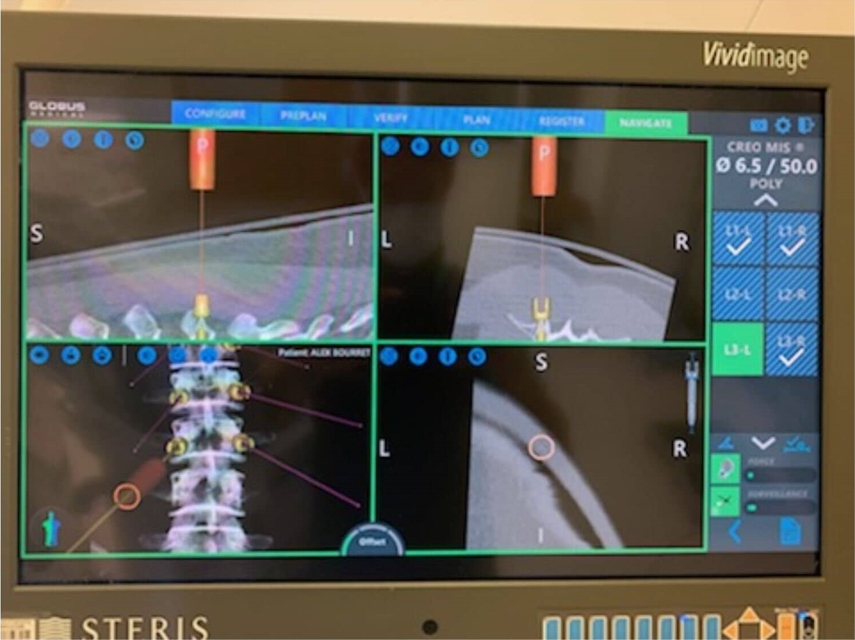
Fig. 3.Placement of a left sided pedicle screw through the Globus Excelsius Robotic Navigation Robotic arm. 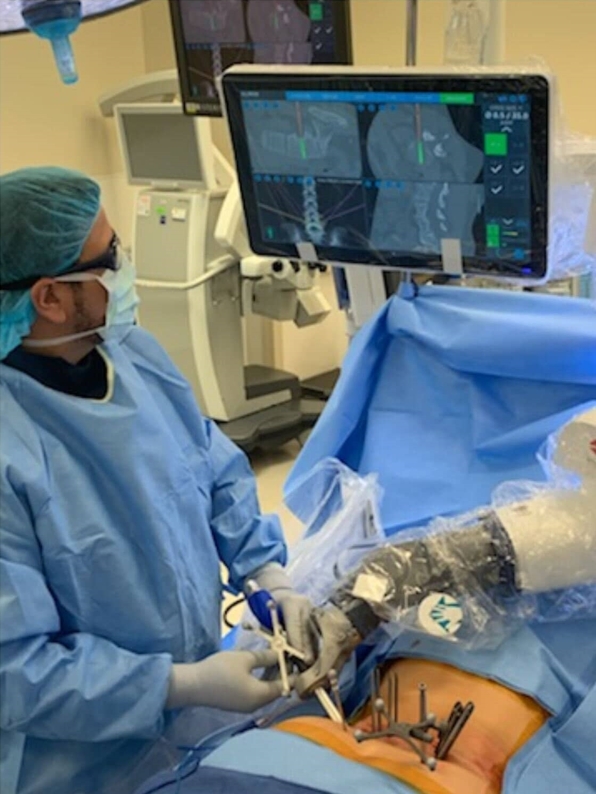
Fig. 4.Bar graph exhibiting differences in EBL, OR time and length of stay between robotic assisted and non-robotic groups. All differences depicted are significant. 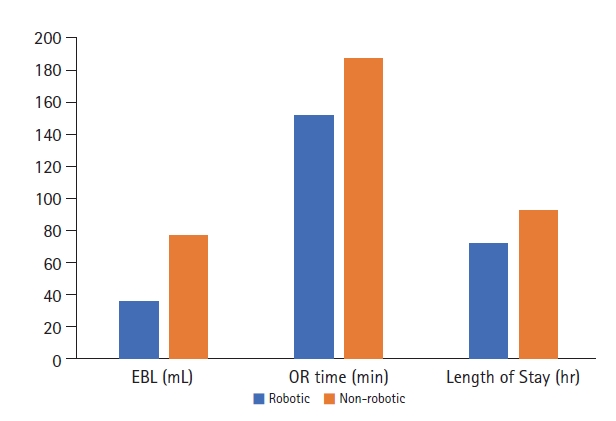
Table 1.Patient demographics and diagnostics Table 2.Surgical comparisons Table 3.Post-operative pain comparisons REFERENCES1. Jaikumar S, Kim DH, Kam AC. History of minimally invasive spine surgery. Neurosurgery 2002;51:S1–S14.
2. Ge DH, Stekas ND, Varlotta CG, Fischer CR, Petrizzo A, Protopsaltis TS, et al. Comparative analysis of two transforaminal lumbar interbody fusion techniques: open TLIF versus Wiltse MIS TLIF. Spine (Phila Pa 1976) 2019;44:E555–E560.
3. Peng CW, Yue WM, Poh SY, Yeo W, Tan SB. Clinical and radiological outcomes of minimally invasive versus open transforaminal lumbar interbody fusion. Spine (Phila Pa 1976) 2009;34:1385–1389.
4. Kim CW. Scientific basis of minimally invasive spine surgery: prevention of multifidus muscle injury during posterior lumbar surgery. Spine (Phila Pa 1976) 2010;35:S281–S286.
5. Mason A, Paulsen R, Babuska JM, Rajpal S, Burneikiene S, Nelson EL, et al. The accuracy of pedicle screw placement using intraoperative image guidance systems. J Neurosurg Spine 2014;20:196–203.
6. Kim HJ, Kang KT, Park SC, Kwon OH, Son J, Chang BS, et al. Biomechanical advantages of robot-assisted pedicle screw fixation in posterior lumbar interbody fusion compared with freehand technique in a prospective randomized controlled trial-perspective for patient-specific finite element analysis. Spine J 2017;17:671–680.
7. Urakov TM, Chang KH, Burks SS, Wang MY. Initial academic experience and learning curve with robotic spine instrumentation. Neurosurg Focus 2017;42:E4.
8. Foley KT, Holly LT, Schwender JD. Minimally invasive lumbar fusion. Spine (Phila Pa 1976) 2003;28:S26–S35.
9. Vaishnav AS, Othman YA, Virk SS, Gang CH, Qureshi SA. Current state of minimally invasive spine surgery. J Spine Surg 2019;5:S2–S10.
10. Marcus NJ, Schmidt FA. Soft tissue: a possible source of pain pre and post minimally invasive spine surgery. Global Spine J 2020;10:137S–142S.
11. Cui GY, Tian W, He D, Xing YG, Liu B, Yuan Q, et al. [Effects of robot-assisted minimally invasive transforaminal lumbar interbody fusion and traditional open surgery in the treatment of lumbar spondylolisthesis]. Zhonghua Wai Ke Za Zhi 2017 55:543–548. Chinese.
12. De Biase G, Gassie K, Garcia D, Abode-Iyamah K, Deen G, Nottmeier E, et al. Perioperative comparison of robotic-assisted versus fluoroscopically guided minimally invasive transforaminal lumbar interbody fusion. World Neurosurg 2021;149:e570–e575.
13. Zhang Q, Han XG, Xu YF, Liu YJ, Liu B, He D, et al. Robot-assisted versus fluoroscopy-guided pedicle screw placement in transforaminal lumbar interbody fusion for lumbar degenerative disease. World Neurosurg 2019;125:e429–e434.
14. Yang JS, He B, Tian F, Liu TJ, Liu P, Zhang JN, et al. Accuracy of robot-assisted percutaneous pedicle screw placement for treatment of lumbar spondylolisthesis: a comparative cohort study. Med Sci Monit 2019;25:2479–2487.
|
|
|||||||||||||||||||||||||||||||||||||||